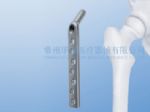
DHS/DCS Plate System (Dynamic Hip Screw / Dynamic Condylar Screw)
The DHS plate system, which has little damage to the soft tissue, offers an effective solution to a variety of intertrochanteric fractures, subtrochanteric fractures, and femoral neck fractures. The DCS plate system is used for the distal femoral fractures and subtrochanteric fractures. Also, it causes little irritation to the soft tissue.
Main Features
1. The low incisura design has greatly lowered the risk of trochanteric bursitis.
2. A wide selection of sizes and cannulas angles provide the versatility to use the system in a variety of clinical circumstance. The cannulas could be the standard or short type.
3. The whole steel plate is devised with numerous screw Agujeros, all of which will not affect the steel plate strength. The fixation points have been increased, on the premise of the relatively small incision.
Indications
DHS
--Intertrochanteric fracture
--Subtrochanteric fracture
--Femoral neck fracture
DCS
-- Intercondylar fracture
--Supracondylar fracture
--Condylar fracture
Materials
Our product is made of Ti6Al4V titanium alloy or 00Cr18Ni14Mo3 stainless steel. Ti6Al4V titanium alloy has been treated with the anodic oxidation surface treatment.
Package and Sterilization Treatment
The product is supplied in the non-sterile package. After originally packed with the paper plastic package, the product will be put into the plastic bag together with the Operation Instructions and the Product Qualification Certificates.
The paper plastic roll bag is used as the inner package. It is compounded from the medical dialysis paper and plastic film. The production environment is controlled with the cleanness of Class 10,000. In the cleaning shop, the package bag is cut according to the product size. Then, its one side will be sealed with the heat-sealing machine. After the bag has been used to contain the product, its other side needs to be sealed with the sealing machine.
The paper plastic, into which the product has been put, will be cleaned in the controlling area where the cleanliness is of Class 10,000. All the cleaning methods have been inspected. The product packed with this method needs no cleaning prior to use. It just needs to be treated with sterilization in accordance with the methods specified in the Product Instructions.
Related Names
Orthopedic Products | Metal Locking Plate | Intertrochanteric Fracture Implant