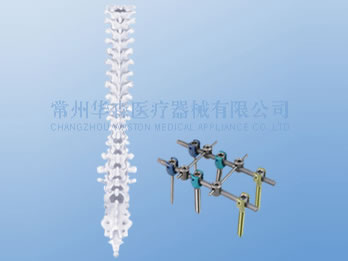
M9 Posterior Thoracolumbar Screw Spinal System
Features
1. Anatomical Reduction
The M9 posterior thoracolumbar screw spinal system is applicable for restoring the normal biomechanical function of the spinal column.
2. Stable Fixation
Stable fixation can speed up the fusion.
3. Preserve the Blood Supply as Much as Possible
The product cultivates a favorable environment for the bone fusion.
4. Painless Early Functional Exercise
It can minimize the damage to the spinal blood supply, nerve, and dura mater. It relieves the pain for the patient, and promotes the function restoration.
Package and Sterilization Treatment
The product is supplied in the non-sterile package. After originally packed with the paper plastic package, the product will be put into the plastic bag together with the Operation Instructions and the Product Qualification Certificates.
The paper plastic roll bag is used as the inner package. It is compounded from the medical dialysis paper and plastic film. The production environment is controlled with the cleanness of Class 10,000. In the cleaning shop, the package bag is cut according to the product size. Then, its one side will be sealed with the heat-sealing machine. After the bag has been used to contain the product, its other side needs to be sealed with the sealing machine.
The paper plastic, into which the product has been put, will be cleaned in the controlling area where the cleanliness is of Class 10,000. All the cleaning methods have been inspected. The product packed with this method needs no cleaning prior to use. It just needs to be treated with sterilization in accordance with the methods specified in the Product Instructions.
Related Names
Lumbar Decompression Product | Low Back Surgery Supply | Back Surgery Plate